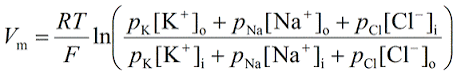|
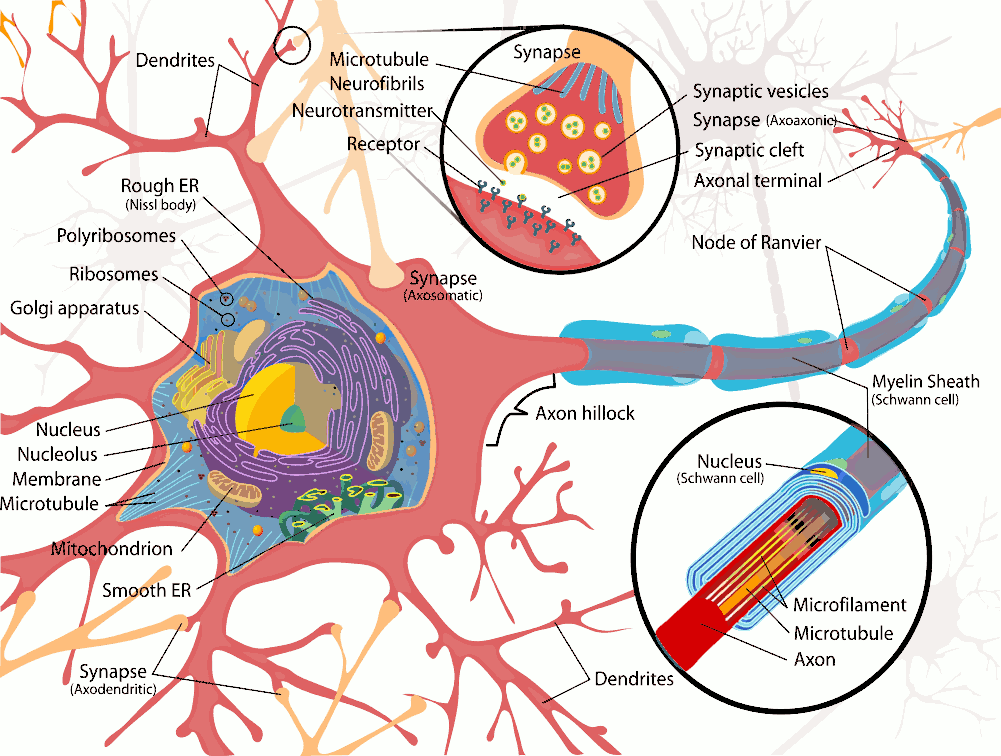
The human body is made up of trillions of cells, of which about 100 billion are nerve cells. Nerve cells or neurons, are specialized to carry electrical signals rapidly over long distances. Neurones have specialised extensions called dendrites and axons which are involved in the collection (dendrites) and distribution (axons) of information within neural networks. Neurones collect and integrate information in the dendrites and cell body (perikaryon) and generate action potentials at the axon hillock. The action potentials pass rapidly along the length of the axon and release neurotransmitters at the axon terminals. Dendrites occur in large numbers, and are branches of the cell body. They receive information from other nerve cells and are not myelinated.
|
|
| AXONS Top |
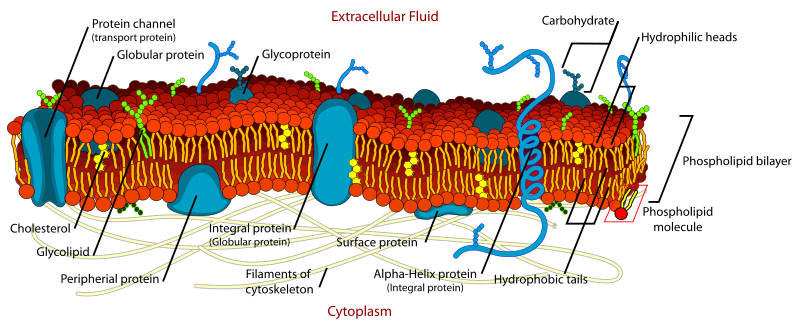
Axons, generally only one per neurone, carry information away from the cell body by means of electrical signals called action potentials that pass along the axonal membrane. This type of signalling is very fast, and is speeded up if the axon has a myelin covering. Axons usually divide into a series of branches, often at some distance from the cell body, and distribute their impulses to other cells (e.g. muscle fibres in the motor unit, or different neurones in the CNS). Axoplasm - the cytoplasm within axons contains high [K+] and low [Na+], compared with Extracellular Fluid (ECF - high [Na+] and low [K+]). These differences in ionic concentration are crucial to one of the main functions of neurones - to conduct action potentials at high speed to the end of the axons. |
Axoplasm also contains neurofilaments and neurotubules which are involved in the carriage of chemicals, vesicles and subcellular organelles to and from the nerve endings by a process called axoplasmic transport. This is a rather slow process compared with the speed of conduction of nerve impulses, and is concerned with the nutrition and connectivity of nerve endings. Substances are transported along neurotubules towards the nerve endings, and this process is called anterograde axoplasmic transport (see below) Axons have specialized structures at their endings called synaptic boutons, which have areas of membrane specifically concerned with the release of neurotransmitters. Neurotransmitters exert their actions on the post-synaptic cell, and can have excitatory or inhibitory effects on that cell, depending on the chemical neurotransmitter (ligand) released at the nerve ending and on the nature of the post-synaptic receptors for this ligand. Post-synaptic cells can also communicate slowly with the pre-synaptic neurone, by releasing chemicals, such as neurotrophins, that are taken up by the nerve ending and transported slowly back to the soma, by a process is called retrograde transport. |
| INTRODUCTION to the ULTRASTRUCTURE of NEURONES Top |
|
Multipolar neurones, such as the motoneurone, can have quite complex dendritic fields, but not nearly as complex as those found in the cortex of the cerebrum and cerebellum. Others, such as the bipolar cells of the retina, are much simpler in structure. The shape of these dendritic fields appears to be due to structural proteins in the neurofilaments that help to maintain the ultrastructure of the neurone. |
The Spanish anatomist, Ramon y Cajal, uncovered some of the complexity of neurones using the Golgi staining method. The cellular theory of the brain was estabished as a result of his investigations: before his time, it was believed that the brain was simply a jelly-like organ. Although neurones show a considerable range of structure, these variations often relate to function, and all neurones with chemical synapses function in the same basic fashion as described below. |
| Variety of Neuronal Structure | Bipolar cells, Cerebellar Purkinje Cells, Cortical Pyramidal Cells |